Scientists may have found a better way to spot early signs of dementia: our eyes
As our body’s central control center, the brain has to keep going at all costs. It makes sense that it’s surrounded by biological safeguards: a bony skull, a sack of tissue, and blood vessels that filter out most infectious bugs. When neurons start to decline—as some do naturally with age—the brain can even quickly rewire networks before we notice anything is amiss.

As our body’s central control center, the brain has to keep going at all costs. It makes sense that it’s surrounded by biological safeguards: a bony skull, a sack of tissue, and blood vessels that filter out most infectious bugs. When neurons start to decline—as some do naturally with age—the brain can even quickly rewire networks before we notice anything is amiss.
But these same safeguards are a huge obstacle for scientists trying to study the brain’s decline—specifically dementia. Grey matter is excellent at adapting to the slow buildup of abnormal proteins, the cause of several forms of dementia, and it lacks the same pain receptors as the rest of our body. So by the time a patient starts becoming forgetful, or having trouble concentrating, the cellular damage is already substantial—and usually irreversible.
In order to slow or prevent dementia, scientists will have to be able to spot it before cognitive systems collapse. Current diagnostic tools for Alzheimer’s, the most common form of dementia, can confirm the build-ups of misshapen amyloid-beta or tau, the hallmarks of the condition. But they are both expensive (PET scans) and painfully invasive (spinal taps). Blood tests are a promising strategy, though after years of research scientists are still struggling to distinguish the chemical signs of normal aging from the signs of dementia.
So a smaller contingent of researchers are trying a novel strategy. They’re looking instead at changes in our sight—a complex sense with several processing steps, each one providing an opportunity for scientists to capture a blip that signals impending doom for the brain. This research is in its infancy. But with the population of aging adults at risk for dementia increasing daily, it’s a critical route of exploration.
The eyes have it
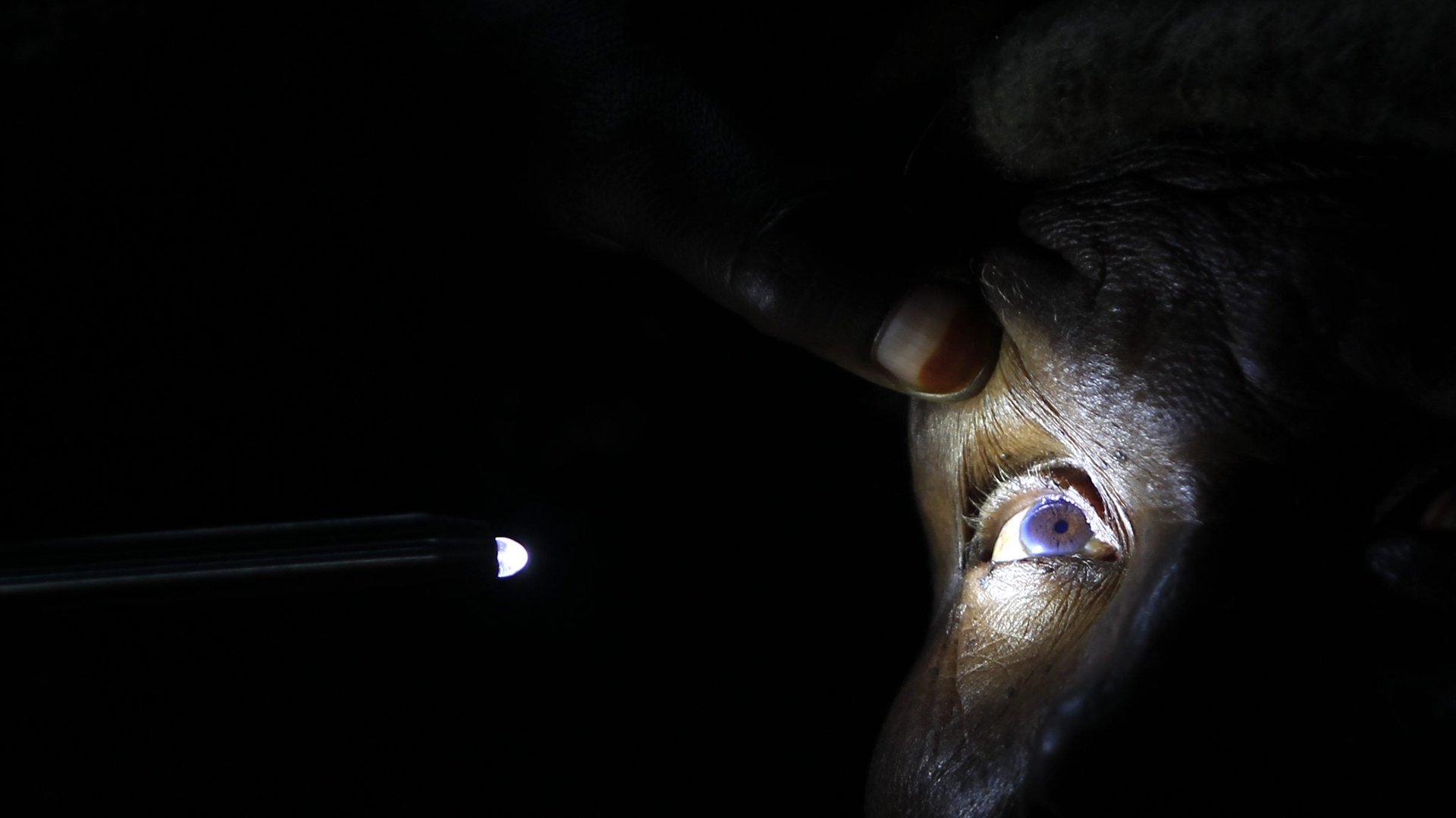
Vision requires our eyes to collect information and send it to the brain, where an entire region works to interpret it in fewer than 120 milliseconds (paywall) by some estimates.
The first stop is the eye—specifically the retina, which picks up images for the brain to interpret. “The retina is a direct extension of your brain,” says Sandra Weintraub, a neurologist at Northwestern University’s School of Medicine. “It actually has neuronal cells.” When something is wrong in the brain, the retina could reflect that damage as inflammation damages tiny blood vessels, called capillaries, around its neuronal cells.
Weintraub thinks that lower capillary counts could help indicate the early signs of dementia. In April of this year, she and her colleague Amani Fawzi, an ophthalmologist also at Northwestern, published work showing noticeable differences in the retinal blood vessels of individuals with mild cognitive impairment or early Alzheimer’s disease and healthy individuals. People with cognitive impairment had fewer capillaries in their retina than those who did not.
It was a small study: just 32 people total, half of them with symptoms of cognitive decline and the others healthy. That’s not enough data to predict if a person is in the early stages of dementia. In order to strengthen this research, Weintraub’s team will next look at a larger group of individuals—preferably with a wider set of ages, and at different stages of progressing dementia.
Making sense of sight
In addition to the eye itself, the brain’s interpretation of images could also serve as an indication of brain health. “The eye isn’t a camera,” says Alyssa Brewer, a neuroscientist at the University of California, Irvine. “The brain fills in all kinds of info based on prior experience and expectation about how things normally look.” This trick likely has an evolutionary purpose: It makes it much easier for us to quickly suss out our environment, and in particular to pick out faces. (This is also why it’s common for us to find faces in objects that are not human, or even animals.)
Changes in the way our brains make sense of the visual world can indicate a larger neurological change. That’s why Brewer studies the brain’s visual cortex, where the bulk of our image processing happens. In particular, she looks at the way regions of the visual cortex are organized into so-called visual field maps. In a small study published in 2014, she and her team used magnetic resonance imaging to compare the visual cortices of a handful of college students, healthy older adults, and two adults of the same age who had been diagnosed with mild Alzheimer’s disease. Although the participants with Alzheimer’s had normal vision, “their maps were completely disordered,” Brewer says. “We were definitely surprised the level of changes that were happening very early on in Alzheimer’s.”
That work measured participants’ visual status quo. By extension, one way to detect early changes in the brain’s visual capacity would be to challenge the system with a hard task—like spotting animals in black and white images flashed for a hundredth of a second on screen.
That’s the idea that London-based start-up Cognetivity is betting on. In January of this year, their research team published work in the journal Nature Scientific Reports showing that their technique could be used as part of cognitive health assessments given in doctors’ offices, although they have yet to be given clearance for clinical use. Tom Sawyer, Cognetivity’s chief operating officer, told Quartz that the company is hoping to run the test on larger populations, including people who have depression or multiple sclerosis, to try to identify subtle changes in visual cognition common among neurological conditions.
Although these are all good leads for better diagnostic tests, they have one problem in common: There’s too much person-to-person variability in eye structure, cortex organization, and visual processing to create a static metric for brain health, the way there is for blood pressure. “We don’t have a good enough handle of what ‘normal’ is in many aspects, even in vision,” says Brewer.
That said, simpler ways of taking snapshots of the brain’s health could make it easier for health care providers to track their patients’ progress over time—and note any seriously concerning dips in cognitive abilities. Even if these tests couldn’t diagnose a specific form of dementia, they could cue health care providers to order tests like PET scans or spinal taps for their patients sooner. Having an earlier diagnosis before their symptoms progress makes it easier for patients and loved ones to plan for the care they’ll need, and it opens the door for them to be enrolled in potentially beneficial clinical trials.