The future of stroke patients may depend on the part-time job of a Canadian surgeon
There are roughly 100 billion neurons in the human brain. These microscopic cells transmit and process information we receive from the outside world and turn our thoughts into action. They are responsible for how we talk, how we move, and how we think. Neurons are, in many ways, what make us us.


There are roughly 100 billion neurons in the human brain. These microscopic cells transmit and process information we receive from the outside world and turn our thoughts into action. They are responsible for how we talk, how we move, and how we think. Neurons are, in many ways, what make us us.
Strokes kill neurons. By starving them of the blood that carries glucose and oxygen, strokes trigger a biochemical cascade that destroys neurons in vast numbers. Ischemic strokes—the most common form, caused by a blocked blood vessel—kill an average of 1.9 million neurons for every minute the patient is untreated. Those dead neurons add up, and in 10 hours, stroke patients can lose as many neurons as they would in 36 years of normal aging.
Roughly 15 million men, women, and children suffer strokes every year, and about half of them are fatal. Stroke is the second-leading killer globally, after its close cousin, heart disease, and far more deadly than cancer and the most life-threatening communicable diseases like AIDS and malaria.
Source: World Health Organization (2016)
Yet despite the enormous toll of stroke, the pharmaceutical industry has been virtually powerless to treat it. After decades spent pouring billions of dollars into the pursuit of drugs to protect neurons after strokes without success, most drug companies abandoned the field by the mid-2000s.
Today, fewer than 5% of all stroke victims worldwide receive any treatment beyond basic palliative care, and the lack of effective stroke drugs remains one of the most glaring unmet needs in medicine.
Stroke is a maddeningly complex problem. The intricacy of the brain, the need for immediate action, and the variability of both strokes and the people who have them make designing and testing drugs an enormous challenge. But the medical establishment has failed stroke patients not just because the research is hard, but because of misaligned incentives, the financial pressures of an industrial drug-development model, and sloppy science.
Stroke nihilism
“Time is brain” is a longtime cliche among stroke professionals, but it’s largely true.
Death follows when a stroke causes the brain to swell, starving it of oxygen, or because the stroke destroys the body’s ability to regulate breathing or blood flow. Others die from complications like pneumonia, which can affect up to one-third of all stroke patients. Stroke weakens the immune system, making it harder for the body to fight lung infections that can occur when stroke victims, who can no longer swallow properly, wind up with food, water, or saliva in their lungs.
For most of history, health workers had no way to help stroke victims. Once a stroke was identified, the patient was made comfortable and family members were given the bad news. Stroke was viewed as a dead end—for patients, for researchers, and for neurologists looking for solutions—and stroke nihilism still permeates the medical establishment.
The bulk of progress in reducing stroke deaths has come from prevention, particularly the introduction of medicines to lower high blood pressure, a leading cause of stroke.
The first—and to date only—medical breakthrough for stroke treatment came when a drug called tissue plasminogen activator (tPA, sold globally under the brand names Activase and Actilyse) was approved by the US Food and Drug Administration (FDA) in 1996.
The goal of tPA is “reperfusion,” the act of returning blood flow to the injured part of the brain. While neurons in the immediate vicinity of the stroke can’t be saved, there’s a larger zone, called the ischemic penumbra, that can be rescued if blood flow can be restored. The longer the penumbra is deprived of blood, the less brain there is to save.
With tPA, emergency-room doctors at last had a way to treat patients. But, as with most things in the world of stroke, there were complications.
In this case, the issue was that there are two kinds of stroke. While the majority (about 85% in the US) are ischemic and caused by a blockage that can potentially be treated with tPA, the rest are hemorrhagic, caused by a ruptured blood vessel, and tPA can be be fatal in these strokes because it prevents the blood from clotting. (For that reason, the drug is also not given to patients on blood thinners or who have other complications. As many as 65% ischemic stroke patients are not eligible for tPA).
Doctors can’t administer tPA without determining the nature of the stroke and that can only be done by examining the brain with a CT scan or some other advanced brain-imaging device. There are less than 4.5 hours after the onset of stroke for doctors to administer the drug—in many cases, not nearly enough time for a patient to get scanned. For patients over 80 or those who had a previous stroke, the window is only three hours.
Read more: With the pharma industry’s repeated failures to develop stroke treatments, stroke doctors have turned to mechanical devices that can clear blocked blood vessels in the brain.
Further, tPA is expensive. The drug, developed by Genentech, has no generic competition, and a 100 milligram vial used in a typical treatment can cost more than $8,300. It also needs to be refrigerated, a challenge for clinics in some parts of the world. As a result, the use of tPA is limited to affluent nations with sophisticated healthcare systems, and even then it is only rarely administered. Since its introduction, tPA has also been plagued by doubts about its safety, stemming from long-standing criticisms of its initial clinical trials. As a result, some doctors won’t prescribe it, even in eligible patients. Fewer than 5% of patients diagnosed with ischemic stroke in the US received the drug, according to a 2014 study. In poorer parts of the world, the number is closer to zero.
Despite tPA’s limited reach, it’s enormously profitable, estimated to make $1.5 billion (pdf) in revenue this year for Roche, the Swiss pharma giant that owns Genentech.
Given tPA’s limitations—and the enormous potential market—researchers have focused on finding a drug that preserves neurons until the brain is reperfused. These drugs, called “neuroprotective agents,” could either save the brain cells in the penumbra until the brain heals, or extend the window of time to preserve neurons in stroke patients until a clot is dissolved by tPA or removed with a mechanical device. In theory, a neuroprotective drug that could be given safely to the 15 million victims of ischemic and hemorrhagic stroke each year—and that could be administered without scanning them first—could generate many times tPA’s revenues.
Jeffrey Saver, a University of California-LA neurologist at the forefront of stroke research for decades, calls neuroprotection the “Holy Grail” of stroke treatment, and like that sacred relic, its pursuit has been an epic tale of frustration and failure.
According to one landmark study, 1,026 potential neuroprotective drugs were tested between 1957 and 2003, in 8,516 separate experiments. Researchers experimented with aged-garlic extracts, uric acid, and compounds engineered from pigs’ brains. Their trials have alluring names, built out of complicated acronyms, that suggest important science is taking place: VENUS, ACTION, SAINT.
None worked.
Those failures cost billions of dollars and wasted the productive years of thousands of scientists. Worse, they salted the ground for future research, ushering in what one researcher called “the nuclear winter” for neuroprotection research. The pharma industry saw more lucrative opportunities elsewhere, and moved on.
“Sadly, the lesson they learned is that they should pursue something else,” says Myron Ginsberg, a neurologist at the University of Miami who has studied the industry’s failures.
But not all scientists accepted that conclusion. On the fringes of industrial medicine, one neurosurgeon has spent the last two decades doggedly developing a neuroprotective agent.
The great white north of neuroscience
The best hope for stroke patients may come not from the giant research labs of industrial pharma, or the biotech hotbeds of Boston or San Francisco, but from the relative backwater of Toronto, Ontario.
Michael Tymianski, now 55, has been working on his drug, called NA-1, since the late 1990s, when researchers were still infused with optimism about developing a stroke treatment. A tall, balding man with a furious work ethic, Tymianski poured himself into developing NA-1 while holding down his day job as a neurosurgeon at a Toronto hospital. His plan was always to develop the drug to the point where it could be tested in humans, then sell it to a pharma company. But no buyers materialized, and eventually Tymianski stopped looking.

Instead, in 2003, Tymianski founded his tiny biotech startup, fueled by confidence in the drug based on his critical insight into brain chemistry.
Following a stroke, neurons become flooded with calcium, which generates free radicals—unstable molecules with an unpaired electron, like nitric oxide, or NO—which causes havoc in neurons. But Tymianski and his team identified a drug that prevents a protein called PSD-95 from binding within neurons, enabling them to resist that lethal accumulation of NO. He called the drug “NA-1,” and, in a nod to the chemical action, named the biotech “NoNO Inc.”
Tymianski speaks carefully and precisely, with a self-deprecation that doesn’t quite mask his underlying self-confidence; it’s clear in our interviews he relishes playing the David to Big Pharma’s Goliath, and is consciously trying to make medical history. More than once he began a sentence, “When the book of NoNO is written…”
He never set out to become a pharmaceutical executive, but with the pharma industry out of the stroke business, Tymianski was forced to go it alone, bootstrapping funding from friends and neighbors. “I was completely unqualified to do it,” he says. “But what was I supposed to do? Put this in a desk drawer and forget about it? I saw this as a natural extension of my obligation as a physician.”
In the 15 years since founding NoNO, Tymianski has raised more than C$120 million (approximately $90.4 million) from about 100 investors, and NoNO is now testing its neuroprotective drug in two ambitious phase III trials in humans.
NA-1 isn’t the first neuroprotective drug to advance to phase III trials, and there are plenty of reasons to be skeptical about its chances for success. As German stroke researcher Ulrich Dirnagl notes, neuroprotection has never been proven to work in humans.
An advocate of more rigorous statistical analysis of stroke research, Dirnagl informally handicaps the odds of various trials for stroke treatments. In an email, he gave his prediction for NA-1: “My guess is somewhere between 10[%] and 20%. Which is the highest rating I have for any running neuroprotection trial.”
Tymianski understands the doubts. But NoNO has learned from the failures of the pharma industry, and has constructed its clinical trials to improve the odds of winning regulatory approval and launching NA-1 into the market. “The road to success is uncharted because everyone before us has failed,” Tymianski says. “We don’t necessarily know what to do to succeed, but we do know what not to do.”
From Chinese food to failure
The history of neuroprotection research begins, strangely enough, with Chinese food.
The flavor additive monosodium glutamate, commonly known as MSG, was first identified in Japan in 1908 by chemist Kikunae Ikeda, who was intrigued by the origins of the umami taste in seaweed. Through products like Ajinomoto, a flavor enhancer, MSG became a staple of Asian cooking.
Chinese restaurants in the US used MSG widely, and it was eventually adopted by western food manufacturers like Campbell’s Soup and sold in American supermarkets, most commonly under the brand name Accent Flavor Enhancer.
Used for decades without incident, MSG began drawing the attention of the medical community in 1968, when the New England Journal of Medicine published a letter to the editor describing symptoms of numbness and palpitations that the writer, Robert Ho Man Kwok, attributed to eating at Chinese restaurants. Kwok, himself a recent Chinese immigrant, identified MSG as a possible culprit. The NEJM dubbed the symptoms “Chinese Restaurant Syndrome” and ignited a long, racially-tinged debate over the health effects of MSG.
The next year, John Olney, a neurologist at Washington University in St. Louis, published a paper in Science demonstrating that in mice dosed with MSG, neurons died, leading to brain lesions. Olney became a crusader against MSG, and enlisted Ralph Nader in a campaign to have the FDA ban the additive. In later years, however, Olney’s conclusions were questioned. The mice in his experiment were injected with, not fed, MSG, and in much higher amounts (relative to body size) than humans ever ingested. The fears over MSG he helped stoke have been largely discredited, but Olney (who died in 2015) had widened understanding of brain chemistry, and how glutamate can kill neurons.

Glutamate is an amino acid found widely in the brain and central nervous system. It lives in the axons—the tendrils at the end of neurons—and plays a critical role in sending messages between those neurons. Once its host neuron sends out the right signal, the glutamate is released, and crosses the synapse, the divide between two neurons. On the other side of the synapse, the glutamate binds to the receptor of another neuron.
Too little glutamate results in a weak signal between neurons, and too much, as Olney discovered in the 1970s, can excite neurons to death by flooding them with calcium in a process he called “excitotoxicity.” Excitotoxicity is believed to be a culprit in a range of neurological diseases such as epilepsy, Parkinson’s, ALS, and stroke.
During a stroke, the loss of blood causes neurons to release glutamate into the spaces between cells. The pooled glutamate over-excites the cell receptors, causing them to open gateways which allow calcium to flow into the neurons. An overload of calcium inside the cells generates free radicals which can damage parts of the cell, like DNA and lipids, eventually killing the neuron. In the 1980s, Steven Rothman, then a neurologist at Washington University, showed in lab experiments this process could be blocked, saving neurons. During this period, research in Germany into the effects of blood deprivation in the brain suggested that neurons could survive and be revived after reperfusion.
Stroke doctors were convinced a major new treatment for stroke was just around the corner, says Dirnagl. “There was huge enthusiasm. Many scientists moved into the field, and industry got very excited. Some people thought they would get a Nobel Prize.”
Mike Tymianski was one of the young researchers swept up in the enthusiasm. “That was indeed a golden time,” he says now. Tymianski graduated medical school from the University of Toronto in 1987 and began training to be a surgeon, but he interrupted his studies to earn a PhD, so he could pursue research while practicing surgery. Neurology offered a particular appeal. “What drove me was curiosity, and the greatest unknowns in the biological sciences were in neuroscience,” he says.
His curiosity has led to some unusual decisions in his life, both professional and personal—like choosing to install his own walk-in wine cellar in his Toronto home. He spent months erecting drywall and installing wiring for lighting, often working between midnight and 3 am as he juggled his other jobs. “If I hire someone to do something, I won’t learn how to do it next time,” he says. “I would never have learned how to hang drywall and run electricity.”
In his (extremely limited) spare time, he’s now building a St. Lawrence skiff, a 20 ft wooden boat, to use at the summer home he and his wife, Dawn, own on Lake Ontario’s Thousand Islands. It is the second marriage for both, and they each brought children into what Dawn, who is CEO of the Nurse Practitioner’s Association of Ontario, calls a “Brady Bunch marriage.” Their combined four children are all now grown.
Through his work as a surgeon, researcher, and now the CEO of one of the few—and one of the most promising—biotech startups in Canada, Tymianski has achieved a place of prominence in the nation’s establishment. He was appointed to the Order of Canada in 2017—among its highest civilian honors—and wears its lapel pin proudly. But he was born in Israel and is a dual citizen.
Poland–>Israel–>Canada
Israel left a powerful impression even though Tymianski moved away while still a boy. “Israel is a high-intensity country,” he says. “I lived through two wars by the time I was 10. It’s hard to forget that.” Growing up during war time may have infused him with a greater sense of urgency, he now speculates.
Tymianski’s father, Marek, was a Polish refugee who fled the Nazis, first to Belarus, then Uzbekistan, before arriving in Israel in 1950, where he met Tymianski’s mother, Rosemary, also from Poland. Most of their extended family was murdered in the concentration camps.
Marek studied to be an engineer, and worked in an Israeli military factory. Rosemary later worked as a lab technician in Canada.
In 1974, the family left Israel and migrated to France, living for a year first in Nice, then Paris, before moving to Ontario, where they had relatives. Tymianski didn’t speak English when he arrived, but spoke enough French to survive in bilingual Canada.
He gravitated towards science in school, and studied math and physics at the University of Toronto, with an eye toward med school. “I didn’t go into medicine with a mission related to my life,” he says. “It’s not that I had a sick sibling or a parent. I just thought it would be a really interesting area to have as a launching pad for future things.”
After med school, as a PhD student in the 1990s, Tymianski was inspired by the new wave of literature exploring the mechanisms behind excitotoxicity and cell death. He began to investigate the role of calcium in neuron death.
Calcium is the “gas pedal” in cells, driving many cellular actions, he says. Cells regulate calcium, but sometimes they fail, and an overload of calcium can be fatal. Through his research, Tymianski found the story was more complicated, at least when it came to brain cells.
When calcium entered through a particular gateway linked to glutamate, called an NMDA receptor, the calcium was toxic, he found. But if it came through another route, the calcium was harmless. Tymianski believed that a protein called PSD-95 (discovered only a few years earlier) was responsible for rendering calcium toxic. But proving it took years of laborious lab experiments.

It was a frustrating process, and Tymianski nearly quit before his graduate student, Rita Sattler, now a professor of neurobiology at the Barrow Neurological Institute in Phoenix, urged him to persevere. Eventually, they proved PSD-95 in NMDA receptors was the key to explaining why glutamate flooded cells with toxic calcium. When PSD-95 was eliminated, the calcium didn’t create nitric oxide, the free radical lethal to neurons. The results of the experiment were published in Science in 1999.
By 2002, Tymianski’s lab had developed NA-1, a drug that links to PSD-95, preventing the protein from binding with the NMDA receptor and eliminating its toxic effects. At least, that’s what he found in lab animals.
Tymianski and his team published these findings in Science in 2002 and, in theory, the pharmaceutical industry should have been lining up outside his door, ready to buy the rights to NA-1, and prove that what worked in lab animals also worked in humans.
But much had changed in the decade since he began his work, and neuroprotection research, once full of promise, was now shunned by the pharmaceutical industry. As Tymianski describes it, stroke research had become a pariah.
A most challenging disease
Traditionally, stroke has been viewed as a disease of the developed world, where fatty diets and tobacco—two leading causes of the condition—are prevalent, and where populations are more likely to live into old age. But because of advances in prevention and improvements in basic hospital care, stroke fatalities have leveled off or declined in affluent nations in recent decades. Meanwhile, as tobacco use and meat consumption have risen in poorer parts of the world, stroke incidence has more than doubled since 1970.
“Stroke is now turning into a tropical disease,” says Derek Lowe, a former pharma scientist who blogs about drug discovery for Science magazine. Malaria and dengue have gone without treatments for decades because their victims are often poor people in poor countries, and pharma saw little opportunity for profit. Stroke now might suffer the same fate.
Though thousands of compounds could potentially act as neuroprotectants, unless a pharma company holds the patent, it has no incentive to pursue them. The US National Institutes of Health, the federal medical science research agency, can’t afford to sponsor thousands of trials. “It really has to be run by industry, and the industry really has to see a profit motive,” Ginsberg says.
There are myriad other reasons that make developing drugs for stroke so difficult.
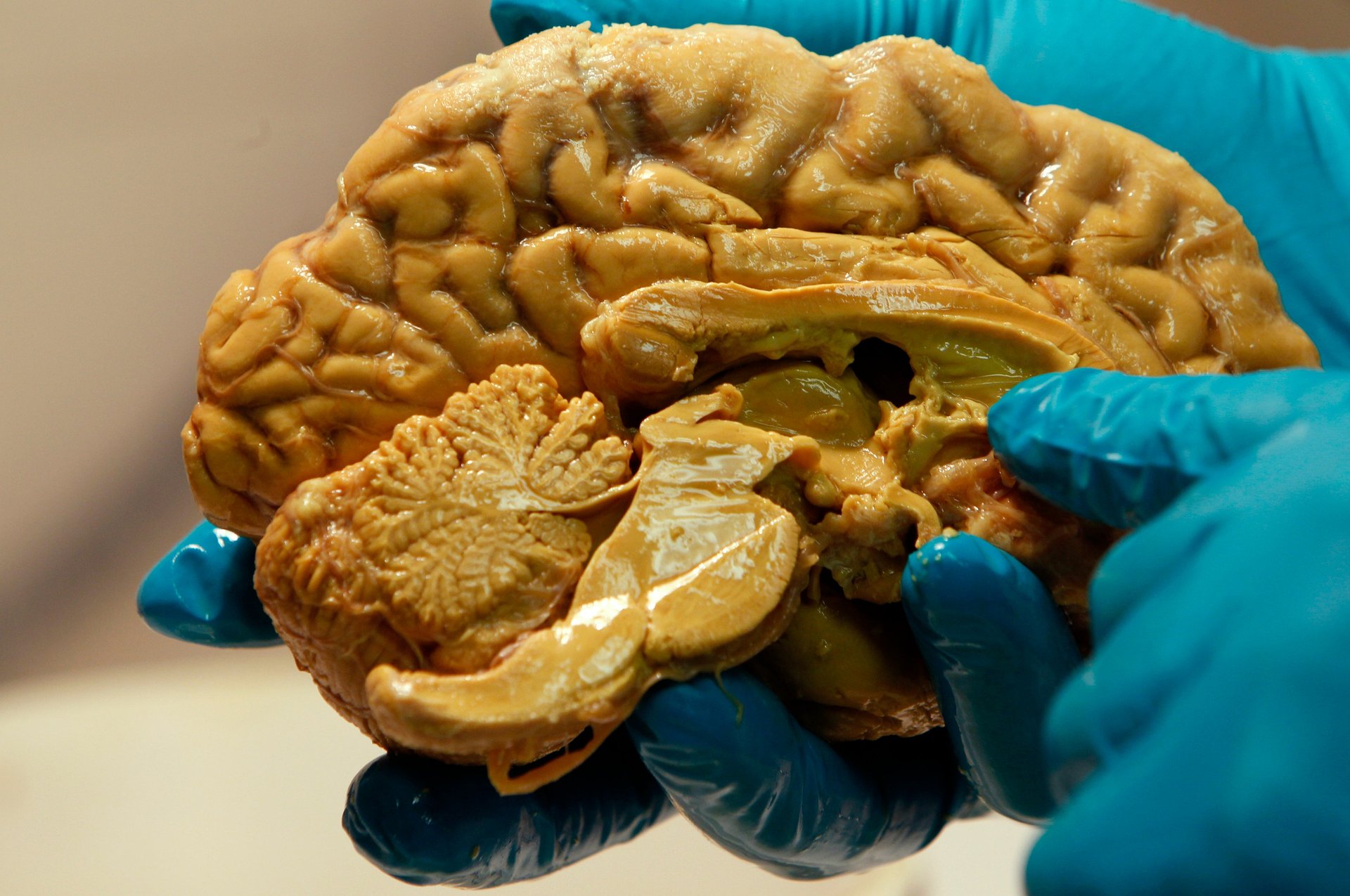
For one, the brain is a particularly challenging organ to work with. Unlike, say, the heart, which is mostly made of uniform cardiac muscle, “the brain is a collection of hundreds of little villages, most of which have limited connection with each other,” says Steven Cramer, a stroke researcher at the University of California-Irvine. “It’s really lots of different organs that kind of grew up together over the last 100 million years.”
That means strokes can have different impacts depending on where they take place; on the size of the blood vessel that is blocked or ruptured; and on the strength of the flow of the collateral circulation system, the network of smaller vessels that can reroute the flow of blood when a major artery is blocked.
Time is brain, but time is not the same for everyone, and depending on the wiring of the vessels, a severe stroke in one patient may not have the same impact in another.
The diversity of human brains, and the wildly varying types of strokes that can affect them, mean identical treatments may produce very different outcomes in clinical trials. Depending on the mix of patients, a trial may indicate a drug is more effective than it actually is, or could be unfairly labeled a failure.
Meanwhile, patient recruitment is a vexing problem for stroke researchers. A stroke is less like cancer or diabetes, where treatment can be planned over months and patients can evaluate their options, than it is like a car crash, where treatment can’t wait. There is little or no time to explain the potential risks of joining a clinical trial, or to secure consent. In any case, patients suffering brain injuries often don’t have the ability to understand what they are being asked to sign. By the time relatives can be found to sign off on enrollment, it may be too late to administer the treatment.
Another reason the pharma industry has struggled with stroke is that outcomes are imprecise. Diseases like cancer lend themselves to definitive answers, such as how long a drug can extend a patient’s life. But stroke treatments are often intended to improve the condition of patients from severely disabled to less-severely disabled, a fuzzy standard that is hard to replicate across all the patients in a clinical trial.
There are also enormous differences in the quality of rehabilitation available to stroke patients in different parts of the world and in different socioeconomic situations. Particularly in the US, where access to healthcare follows income, poor patients may receive inadequate rehabilitation, and therefore won’t show the same improvement as a wealthy patient. It means the results of clinical trials can be warped by factors as unpredictable as the patient’s health insurance and access to rehabilitation facilities.
There are financial obstacles to R&D as well. Increasingly, large pharma companies have outsourced innovation to small, nimble biotech startups founded on the strength of one or two bright ideas. The venture capital firms that bankroll those startups only make investments where they think there will be a lucrative exit, usually in the form of an acquisition, and so only place bets on drugs that target an in-demand therapy area, says Bernard Munos, a fellow at the Milken Institute who once worked for Eli Lilly & Co. and now consults for the pharma industry.
As stroke-research failures multiplied and the pharma industry walked away, VCs noticed. “VCs are very sensitive to what diseases are hot, because they’re thinking about the exits,” Munos says. “No one is saying ‘invest in stroke research.”
But ultimately, it’s the want of a great idea, not market incentives, that has been the biggest barrier, Lowe says.
“More money would probably help a little bit, but the problem is ideas,” he says. “There is a ton of money, an unbelievable amount of money just sitting there for anyone who can do anything about Alzheimer’s. Have we done anything about Alzheimer’s? We have not.”
Rodents and primates
After the initial waves of discovery into the role of glutamate in neuron death, researchers began experimenting with compounds that targeted the NMDA receptors that controlled the flow of glutamate into the cell. Many drugs showed promise in lab animals, only to fail in human studies, occasionally causing serious side effects. Often, the drugs would effectively disrupt the NMDA glutamate receptors, but would cause havoc throughout the rest of the nervous system.
In retrospect, there were serious flaws with the animal trials, says Ashfaq Shuaib, a professor of neurology at the University of Alberta. To save money, laboratories would use smaller mammals, like rodents, cats, or dogs, instead of primates whose brains more closely resemble those of humans but which are significantly more expensive. The rodents were often inbred, so they were genetically similar, and young and healthy. Researchers induced strokes in the animals by injecting a clot into the middle cerebral artery of their brains, so each animal suffered the same size and type of stroke.
This systematic approach made the results more uniform and the data cleaner, but it didn’t really resemble the variety of stroke in humans. The animal models presented researchers with unrealistically strong signals, Shuaib says. Further, the animals were given large doses of the drugs, much larger than what would be given to human patients. “It was quite naive,” he says now.
As failures in neuroprotection-drug trials began to mount, academic and industry researchers convened in March of 1999 to examine their errors and adopt best practices for future trials. The Stroke Treatment Academic Industry Roundtable (or STAIR), produced a number of guidelines for researchers (pdf), such as avoiding the use of mice and gerbils in animal trials, and ensuring experiments were successfully replicated in two or more laboratories before advancing to human patients.
But while the STAIR guidelines added rigor, they still couldn’t control for human error.
The sad story of SAINT
In the mid-1990s, AstraZeneca, the UK pharma giant, struck a deal with Centaur Pharmaceutical, a biotech startup, for the rights to develop Cerovive, a neuroprotective drug candidate. The drug, also known as NXY-059, showed promise in animal and early-stage human trials and by the early 2000s, enthusiasm was high even as the rest of the industry was shunning neuroprotection.
In a complex undertaking between May 2003 and November 2004, AstraZeneca enrolled 1,722 patients, across 158 hospitals in 24 countries, in a trial for Cerovive called SAINT. The trial adhered to the STAIR criteria: Patients could be given the drug up to six hours after their stroke, but each hospital was required to maintain an average of four hours. The six-hour limit “was picked out of convenience, rather than evidence, to get as many patients as quickly as possible, as fast as possible,” says Shuaib, who was one of the authors of the study. “It was a mistake.”
“The push from pharma was ‘get the study completed quickly,’ so unfortunately what that meant was you put it into all kinds of patients,” he says.
The results of SAINT, published in February 2006, showed a modest benefit for patients who were given Cerovive. Still, in the world of neuroprotection, even a small sign of success was considered significant; investors and the financial press took notice, and stroke researchers celebrated the results.
In retrospect, researchers should have been more skeptical about the SAINT results, according to Shuaib. “There was a positive sign, but it was just on the margins,” Shuaib says. “It met the criteria set out by the statisticians so it was good enough for all of us.”
To satisfy the FDA’s requirements for two completed phase III trials, AstraZeneca had simultaneously launched an even bigger trial, SAINT II, which was planned to include 1,700 patients. But because of the inconclusive results of SANT I, AstraZeneca doubled the size of SAINT II, enrolling 3,306 patients from 362 hospitals in 31 countries.
On Oct. 26, 2006, AstraZeneca released a press release that stunned the medical and financial worlds: Based on the results of SAINT II, Cerovive was a failure.
AstraZeneca’s stock fell 7.7%. Shares of Renovis—a biotech that bought Centaur in 2002—plunged 76%, and it laid off nearly half its workforce the following year. By 2009, Renovis was no more.
The blow to stroke research was longer lasting.
John Patterson, AstraZeneca’s head of drug development, declared defeat not just for Cerovive but for the entire class of drugs to which it belonged. “We don’t think there is any mileage in a neuroprotectant approach,” he told reporters in a conference call the day the failure was announced, Reuters reported. “We’ll not look to acquire any projects in the stroke area in this particular field… we think we’ve done the definitive study here.”
AstraZeneca declined to discuss its experience with Cerovive.
While the SAINT trials may have been the most spectacular failure, throughout the history of neuroprotection investigation, researchers let their optimism get in the way of sound science, says UCLA’s Jeff Saver. Scientists didn’t rigorously blind and randomize animal subjects, and they didn’t declare their metrics for success before the trial, instead cherry-picking the results that made the best case for the drug’s effectiveness.
Saver calls the history of neuroprotection “chastening.” There were no villains, he says, and plenty of blame to go around. “It was an embarrassment but not a scandal,” he says. “Everyone was trying to do right, but we weren’t smart enough to recognize it.”
NoNO for an answer
Tymianski, now the CEO of his own drug company, is philosophical about the failings of the pharmaceutical industry. It’s easy to find mistakes in hindsight, he says. “It’s harder for me to judge what happened now that I’m walking in their shoes.” Still, “there’s no question, in retrospect, you can spot the errors,” he says. He remains astonished that Cerovive could proceed as far as it did with no real evidence it worked. One reason Tymianski chose to form NoNO and develop the drug himself, he says, is “I didn’t trust [pharmaceutical companies] to do the right thing scientifically.”
But even if he was a fan of the drug industry, it’s not like pharma had any interest in NA-1.
After the publication of the 2002 Science paper introducing the drug, there was no stampede to buy NA-1. As Tymianski considered future directions, he reached out to potential mentors, including John Evans, a cardiologist who had chaired Allelix Biopharmaceuticals, one of Canada’s first biotechs. “He said, ‘There’s no such thing as a nice drug company,’ and, ‘stay away from VCs for as long as you can,’” Tymianski recalls.

Tymianski began approaching investors, starting with friends and family. He was his usual blunt self. “The pitch was ‘Kiss your money goodbye. There’s no exit strategy and you’ll probably never see it again. But if we win, our patients will win and you will do fine,’” he says.
To his surprise, most of the potential investors he asked have signed on.
One early supporter was Josh Josephson, the owner of a chain of Canadian optician offices, and Tymianski’s neighbor. Others followed, including Ron Kimel, a Toronto real estate mogul, and Kevin O’Leary, a Canadian entrepreneur better known to Americans as “Mr. Wonderful” from his appearances on the TV show Shark Tank. Not all of NoNO’s investors are wealthy, Tymianski says, and they all have his mobile number. Tymianski interrupted one of our conversations to take a call from an investor.
Most of his initial investors have stood by him, Tymianski says, and he’s just completed a seventh round of angel investing. NoNO has also secured C$20 million in grants, including more than C$6 million from the Brain Canada Foundation to fund phase III testing. (Tymianski calls himself a “silver-backed gorilla” when it comes to winning grants.)
Because NoNO has made it this far without venture capital or corporate investment, Tymianski has the freedom to develop NA-1 as he sees fit, without pursuing the shortcuts that proved the downfall of other neuroprotection candidates.
For example, NA-1 was tested by independent researchers in multiple centers, to ward against biased findings from Tymianski’s lab. In addition, after it was found to work in rats, NA-1 was given to long-tailed macaques, a monkey species chosen because of its anatomical similarities to humans.
Tymianski also developed an innovative approach to the core challenge that pharma had encountered in human trials: patient recruitment. He did it bypassing stroke patients altogether in his phase II trial.
In his day job as a neurosurgeon, Tymianski often treats brain aneurysms, which are caused by a weak area in blood vessels bulging or ballooning. In the course of repairing these aneurysms with mechanical devices, small ischemic strokes can be caused by dislodged cholesterol and blood clots blocking minute arteries. These small strokes aren’t life-threatening and are predictable and visible through MRI scanning. So Tymianski reasoned that he would have a better chance of proving NA-1 penetrated brain cells in a population of aneurysm-repair patients than trying it in a much more diverse group of ischemic stroke patients who arrived in emergency rooms.
Over two and a half years, NA-1 or a placebo was administered to 185 patients undergoing brain-aneurysm repair surgery at 14 hospitals in the US and Canada, in a phase II trial called ENACT. The results, published in The Lancet Neurology in 2012, showed patients treated with NA-1 had fewer strokes, and a subset of the aneurysm patients that suffered hemorrhagic strokes showed improved neurological functions compared to the placebo group after 30 days.
ENACT, FRONTIER and ESCAPE NA-1
In the small world of stroke research, Tymianski’s work has drawn attention because of the creativity of his approach and the promise of his results. “It was developed in a thoughtful way,” says Ginsberg. Based on the success of ENACT, NA-1 is now being tested in two phase III trials, with the first results expected in early 2020. If they are successful—pending regulatory approval—stroke doctors may at last have a drug that can save brain cells.
One trial, called FRONTIER, is administering NA-1 to stroke victims in ambulances, so it gets into patients as quickly as possible. So far, more than 300 patients have received NA-1 or the placebo, in a median time of 59 minutes after their stroke.
Another trial, called ESCAPE NA-1, is an effort to see if NA-1 can help ischemic stroke patients who have been reperfused with mechanical devices. The trial is being conducted at nearly 50 stroke centers around the world.
Together, Tymianski says, the trials are an attempt to replicate the conditions where he knows NA-1 works: in lab monkeys given the drug soon after strokes are induced, and where blood is restored to the penumbra. Unlike in the trials for Cerovive and other experimental neuroprotectants, where the drug went from the carefully controlled environment of the lab to the chaotic emergency rooms of stroke care, NA-1 is being tested in humans in situations designed to give it the best chance of showing results and winning regulatory approval.

If successful, Tymianski will need to decide what to do with NA-1. Options include an outright sale of NoNO to a pharma company, licensing the drug, or forming a partnership. Or NoNO could try to commercialize the drug itself, perhaps by filing an IPO. “We will do what’s best for the company, and for the people that it serves,” Tymianski says.
Maintaining control of NA-1 would give NoNO some say in its pricing, and availability in the developing world, where it could potentially be the first stroke treatment of any kind.
Because NA-1 doesn’t operate in blood vessels like tPA, the hope is that it can be safely administered to victims of both hemorrhagic and ischemic strokes, obviating the need to CT scan patients first. The drug could therefore be given to stroke patients in rural settings or poor countries where the medical infrastructure required to administer tPA is unavailable.
Tymianski says he’d like to be able to offer NA-1 at cost in countries where most people couldn’t afford even a modest price. “There is no profit to be made from people who have no money,” he says. “Why not be a good guy?”
The high cost of failure
Prescription drug prices in the US have risen dramatically in recent decades: 57% from 2006 to 2014. The pharma industry blames the growing list prices on the costs of developing a successful drug. And drug discovery is certainly expensive: A recent study places the cost at $2.6 billion for a single approved medicine. That’s a lot for a pharma company to make up in the 20 years of a drug’s patent-protected life.
But that price tag doesn’t just include the cost of originating a successful drug; it also folds in the R&D costs of the many more unsuccessful attempts. Drug companies can’t endlessly absorb those losses, so they are passed along to consumers in higher overall drug prices.
Failure is part of science. It’s build into the most basic concept of trial and error that underpins the scientific method. We need to tolerate failure—and pay for it—in order to benefit from the successes. But the expectation is that failure is the result of honest attempts, based on the best information available, using the most rigorous and sophisticated methodology.
When the drug industry wastes billions of dollars because of sloppy science, we are quite literally paying for its mistakes.
The fact that the prognosis of millions of future stroke patients may rest on the part-time job of a Canadian surgeon speaks to how badly broken stroke research has become.
At the institutional level, stroke is an unwanted step-child. A disease of both the blood system and the brain, it falls between the cracks of cardiology and neurology, embraced by neither discipline. The neglect is apparent in the funding: While research into lung disease and Alzheimer’s each received almost $2 billion in federal funding from the National Institutes of Health in the 2018 fiscal year, stroke research was given just $349 million.
Stroke has few advocates. Its victims are often already old and infirm, and its survivors are ill-equipped to hold rallies and lobby their legislators. There is no ice-bucket challenge for stroke.
Many of the approximately 25 million survivors of stroke living around the world today have serious impairments, some permanent, that tax resources for families and healthcare systems. Stroke costs the planet 113 million disability-adjusted life years annually (a metric that includes the impact of early deaths and total years lived with disability). In the US alone, stroke cost $34 billion in medical expenses and lost economic activity in 2013, the most recent year for which data are available.
Despite the pharma industry’s abdication, neuroprotection is a big enough problem—and represents a big enough jackpot for the researcher that solves it—that Tymianski isn’t alone in trying. Treatments in clinical trials include a drug that increases the supply of oxygen to the ischemic penumbra and a stem-cell therapy that promotes healing in damaged cells. Other studies are experimenting with nitroglycerin patches to control blood pressure in stroke patients, and drugs to reduce brain swelling.
In a world where very little is known to work, very little can be ruled out. There is a long tradition of medical research advancing through blunders and happenstance —Alexander Fleming famously discovered penicillin by accident. It may be that stroke’s best hope is a result of the industry ignoring Tymianski when he first discovered NA-1, allowing him to develop it free of their meddling
As a global society, we tackle diseases not in the order of greatest need, but according to the priorities of the pharmaceutical industry. Their decisions are shaped by the potential return on investment, and a distorted regulatory structure that can make a treatment for a rare disease more profitable than one for a massive killer.
On occasion, the needs of the drug industry and society line up, and we have a bounty of new cancer drugs as a result. But when the research is hard and the patients are poor, the market fails. We can hope for people like Mike Tymianski to provide cures, but we shouldn’t have to count on it.