Table of contents
A new era of medicine | Glossary | Making genes into medicine | A brief history of big pharma | What genetic medicine could treat | How genetic medicine can live up to its promise
A new era of medicine
Chances are, you’ve never heard of Pieter Cullis. But if you’ve received the Pfizer-BioNTech or Moderna Covid-19 vaccines, you have him, in part, to thank.
Cullis is a biochemist at the University of British Columbia in Vancouver, Canada. He’s dedicated more than four decades of his life to figuring out how to package and deliver the building blocks of genetic code inside a tiny, specialized particle to help treat or prevent diseases. One of his biggest achievements came in 2018, when the US Food and Drug Administration (FDA) approved Onpattro, a drug for a rare genetic disease, which uses encasing technology from his company Acuitas. Onpattro quiets the problematic gene responsible for the condition; to get it where it needs to function in the body, it’s packaged in a lipid nanoparticle that originated in Cullis’ lab.
The drug helped people, but didn’t seem to portend a medical revolution. “[Onpattro is] something that only applies to 50,000 [patients] worldwide,” he says. But less than four years later, lipid nanoparticles exceedingly similar to the ones used in Onpattro are being delivered to billions of people across the globe to prevent serious infections of Covid-19. The German drug company BioNTech, in partnership with Pfizer and Acuitas, developed a Covid-19 vaccine that uses mRNA, short for messenger RNA, which instructs our cells to make a new protein—in this case, the spike protein on the SARS-CoV-2 virus—so our immune systems respond to it like a threat. Cullis’ company Acuitas Therapeutics licensed lipid nanoparticle packaging to transport and protect the mRNA to BioNTech. The Moderna vaccine uses a similar approach, and while its lipid nanoparticles are not from Acuitas, they’re close cousins.
As far as genetic-based medicine is concerned, this is just the beginning. “It’s a bit of a gold rush at the moment,” Cullis says. “Pretty much any disease you can name, you can go after with an mRNA approach.”
The Pfizer-BioNTech and Moderna vaccines are ushering in a new era of medicine. After decades of research—and a Herculean effort to address an ongoing pandemic—new pharmaceutical companies are ready to roll out novel therapies by tweaking the way our bodies use genetic code. Through these new approaches, drug developers can direct our bodies to make beneficial proteins or halt the production of toxic ones.
In theory, there’s no limit to the kinds of diseases we could treat this way. “It’s revolutionary,” Cullis says. “We’re starting to use the biology that our bodies use in a therapeutic way.”
At the moment, however, these treatments are not widespread and generally not accessible for most common diseases. To get them there, researchers and biotech companies will have to perfect the drug delivery systems to plant each genetic therapy in exactly the right place—the last 10 kilometers in a marathon of research. The finish line is just beyond reach, but well attainable in the near future.
“Twenty years from now you’ll be taking a lot of these for whatever ails you,” says Steven Dowdy, a cancer biologist at the University of California at San Diego.

Glossary
This new wave of medicine is built around our bodies’ genetic code. To understand this exciting field, it helps to get a sense of the terms that make up its foundation. They might remind you of high school biology, but with an updated twist. We’ll also remind you of these terms where needed in this piece.
DNA: DNA, or deoxyribonucleic acid if you’re feeling fancy, is the master copy of biological information that makes us us. It’s made of nucleic acids and is tucked away safely in the membrane-protected nucleus of every one of our cells.
DNA is the blueprint our cells follow as they make proteins. DNA’s double-helix structure makes it harder for copying errors, which could cause diseases, to slip through.
Genes: Sections of DNA that code for proteins. Genes also have regions called enhancers and silencers that help our cells decide how much of a protein to make, and when to stop.
Gene therapy: Any form of medicine that modifies a person’s genes. This could involve editing some of their cells to attack cancerous cells, or injecting an entirely new gene into a specific part of the body to replace one that’s missing or broken. Typically, gene therapy is focused on introducing changes to our DNA—either by putting new DNA to our cells, or actually altering cells’ genetic code.
One of the original forms of gene therapy is to introduce a snippet of DNA that codes for a functional gene into a cell where the existing one is broken. This new bit of DNA can be delivered with a virus that’s been modified to carry the new DNA—that’s how Luxturna, made by Spark Therapeutics, corrects a rare form of childhood blindness. This technique is also the basis for the Oxford/AstraZeneca Covid-19 vaccine.
RNA: Ribonucleic acid is a single-stranded form of genetic material. Like DNA, it’s made of nucleic acids. Some organisms, like the SARS-CoV-2 virus that causes Covid-19, only have RNA as their genetic code.
Humans use RNA, too, though it is not our main blueprint for life. We have several types in our cells to turn our DNA into proteins. Some RNA copies down instructions from DNA in the nucleus and ferries it out to the rest of the cell. Other RNA forms the walls of the protein factory, while another assembles the building blocks of proteins in just the right order so the finished product can perfectly do its job.
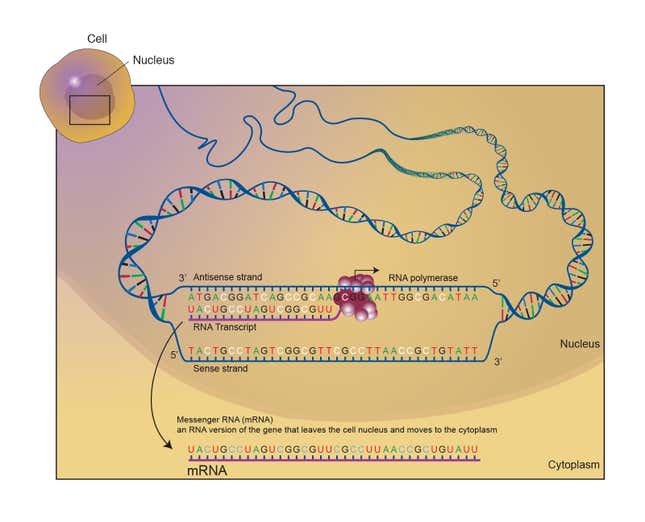
Precision genetic medicine: Any kind of therapy or vaccine that uses nucleic acids to give our cells recipes for new proteins, or turning off the production of a protein that is toxic.
mRNA: Messenger RNA is a type of RNA that copies short snippets of DNA from the nucleus and carries it to ribosomes, where proteins are made. Some of these proteins carry out jobs within the cell, and some of them get carried out beyond the cell. Being alive and healthy depends on our ability to make and later break down proteins (a job that’s carried out by—guess what?—other proteins).
mRNA doesn’t have to come from our own DNA. Scientists have figured out that they can introduce external mRNA into our cells to produce new proteins our bodies aren’t making on their own. This is the basis for the Moderna and Pfizer-BioNTech Covid-19 vaccines.
RNAi: RNA interference neutralizes mRNA so it can’t form a protein. Short interfering RNA (siRNA) and microRNA are two types of RNAi.
RNAis are useful for medicine because they can, for example, treat a genetic disorder in which a person’s body produces a faulty copy of a protein that makes them sick. Cullis’ Onpattro uses siRNA to quiet a gene that forms a toxic, deformed protein in patients with an inherited condition called hereditary transthyretin-mediated amyloidosis.
ASO: Antisense oligonucleotides are lab-made nucleic acids that correct mistakes mRNA can make as it pulls together protein recipes from our DNA (a process called “splicing”). They can also turn off mRNA to block the protein from being made. RNAis can be ASOs, but they don’t make up the entirety of the category, the way spaghetti is only one kind of pasta.
Lipid nanoparticles (LNPs): Lipid nanoparticles are lab-made fatty vesicles that mimic the structures that transport goods in and out of our cells on a daily basis. They can either have a liquid center to transport individual drugs, or they can be a blob that binds to genetic material to carry it into our cells.
Ligands: When attached to lipid nanoparticles, they act as protein labels that work like shipping addresses.
Endosome: The cell’s holding area for incoming cellular cargo, which are proteins or fats and sometimes foreign nucleic acids, like precision genetic medicine. An endosome assesses where in the cell new goods need to go, and then releases them through its lipid membrane to their destination.

Making genes into medicine
Now that mRNA vaccines1 are in millions of arms worldwide, some experts have argued that pharmaceuticals are now in their fourth wave (the first wave was mass-producing chemicals from plants or bacteria; the second was lab-made compounds; and the third was making molecules similar to those found naturally in our bodies).
It’s the culmination of decades of work across biology, medicine, chemistry, and physics. Ever since scientists discovered DNA in the 1950s, others have been trying to hack it to treat diseases. They do so in a few different ways. Sometimes they can extract certain cells, modify them with gene editing enzymes like Crispr, and then reintroduce them to our bodies. Other times, they can insert a new gene to make up for a faulty one using a benign, modified virus to sneak into our cells. These are called gene therapies, and they were some of the earliest iterations of genetic medicine.
Each of these methods presents its own challenges. For one thing, using Crispr with a person’s body could cause an immune reaction, which could potentially cause more damage than the problem the treatment was trying to solve. For another, some scientists and patients balk at the idea of permanently changing our genetic master code in select cells. Even if it works, it’s a cumbersome process, and if a mistake happens, it’d be permanent for the cell’s lifetime (although notable, these therapies could never jump to non-targeted cells). The approved gene therapies are only suitable for people who have tried other medications and have no other options.
RNA2 and other kinds of lab-assembled nucleic acids are appealing because they can influence the ways our cells make proteins from DNA—leaving the master copy alone. But scientists run into delivery problems within the body: It’s hard to get nucleic acids to reach their respective targets.
One reason was that, compared to other kinds of medicines, nucleic acids3 are big. The amount of genetic material needed to tell our cells to make a certain protein, or to change the way they make existing proteins, is about 25 times as big as the small molecule drugs that could passively sift their way across our cell membranes, says Dowdy, who studies nanoparticle delivery for chemotherapies.
And size aside, “you can’t deliver naked mRNA to cells,” says Dowdy. Our bodies are on high alert for any kind of random genetic material floating around because it’s usually the sign of an unwelcome pathogen. In fact, our bodies secrete copious amounts of the enzyme ribonuclease specifically to break up the single strands of genetic material usually associated with diseases. And though that enzyme probably protects billions of people from various illnesses each year, it means that mRNA and other introduced genetic material is pretty unstable in our bodies on its own.
Delivering drugs using modified viruses, as scientists have done with gene therapies4, works, but with a catch: Though viruses are skilled cellular invaders, eventually our immune system wisens up and attacks them like they would any pathogen. This defense mechanism, as helpful as it’s been in human evolution, thwarts the ability of the virus to deliver its therapeutic goods.
Thankfully, chemists came to the rescue. Lipid nanoparticles5, which had now been in the works for nearly 50 years to improve all kinds of drug delivery, were the obvious choice for packaging nucleic acids so they could be safely introduced into the body and then slip into cells. Because these particles mimicked naturally-occurring lipid vesicles already in our bodies, they’d be able to arrive at the desired cell ready to use. “It’s taken 50 years of chemistry,” says Dowdy. “The entire field of RNA therapeutics stands on [chemists’] shoulders.”
The first clear success, a form of precision genetic medicine cleared for medical use in the US, was an ASO6 called fomivirsen, which was made by Novartis to treat a rare form of eye inflammation in people with compromised immune systems. That happened in 1998. “Then [the field] crashed,” says Chris Anzalone, the chief executive officer of Arrowhead pharmaceuticals based in Pasadena, California. Demand for fomivirsen plummeted after other antiretroviral therapies took off, so Novartis pulled the plug. There wouldn’t be another precision genetic treatment approved by the FDA until 2016.
In the 23 years in between, scientists continued to tackle nucleic acids’ delivery problem—albeit largely separate from the field of big pharma. Drug companies at the time were either focused on developing blockbuster small molecule drugs, which require scientists to have a profound expertise in the exact ways diseases work, or the early iteration of biologics—drugs based on molecules that already exist in our bodies, like insulin or antibodies.
Instead, most of the work came from two other sources: academics at universities or other research organizations who could afford to study something that wouldn’t have immediate clinical applications, or the tiny, private biotech companies spun off by universities. These research teams developed entire libraries of lipid nanoparticles that could be paired with different kinds of nucleic acids. They also worked on developing ways to keep these nucleic acids more stable.
These smaller startups didn’t have the same kind of financial stability as larger pharmaceutical companies or the in-depth knowledge of diseases required to make small molecule drugs. But their research had so many therapeutic applications that new companies were primed and ready to license their work to others—or simply get bought by them.
Since 2010, the number of small biotech companies focused on delivering genetic medicine has boomed. Globally, there are hundreds of startups that have popped up in recent years, focused on manufacturing different kinds of genetic code, delivery systems to get them there, or a combination of both.

A brief history of big pharma
1668: Darmstadt, a pharmacy in Germany, is founded. In the 1800s, it went on to become the pharmaceutical company Merck. The companies that would become several of today’s pharmaceutical giants, including GlaxoSmithKlein (its parent company, Plough Court Pharmacy, was founded in 1715), Pfizer (1849), and Bristol Myers Squibb (1887), were founded in the 1700 and 1800s. During that time period, most drugs were the result of finding active ingredients of drugs in plants via medicinal chemists, which is known as the first wave of pharmaceuticals.
Early 1900s: Bayer starts mass producing aspirin. It’s one of the most successful commercial drugs, but it’s competing with things like cod liver oil during a time when drugs are largely unregulated. The discovery of penicillin comes in 1928.
1953: UK-based biologists John Watson, Francis Crick, Maurice Wilkins, and Rosalind Franklin uncover the double helix structure of DNA. They won the Nobel prize in medicine for their work in 1962, although Franklin was omitted.
1962: US Food and Drug Administration requires drug companies to provide clinical trial data proving their drugs are safe and effective.
1965: Robert William Holley, Har Gobind Khorana, and Marshall Warren Nirenberg become the first to sequence genetic material—a form of RNA. They win the Nobel Prize for their work in 1968.
1965: Alec Bangham, a biophysicist and clinician University College London, comes up with fatty vesicles that could theoretically be a delivery system for drugs. He called them “multilamellar smectic mesophases,” but others nicknamed them “banghasomes.”
1970s: The drug industry focuses on developing “blockbuster” drugs. These drugs—which technically bring in more than $1 billion in sales—are all based on small molecules. They include things like Valium, the contraceptive pill, and chemotherapies. Small molecule drugs like these were often referred to the “second wave” of pharmaceuticals
Also 1970s: Contract research organizations, or CROs, hit their stride. These companies are essentially the sous chefs to research groups or pharmaceutical companies trying to launch a new drug product. They offer to fill in the gaps that their biomedical client needs. In their early days, these companies primarily functioned to do the complex number-crunching drug companies needed to either find new candidates for drugs or analyze their results. As computer processing improved, CROs started venturing manufacturing products, too.
1971: Cetus, the first biotech company, is established in Berkeley, California, working on components of antibiotics. It doesn’t become a household name (it’s now a part of Novartis) but it’s the first of the third wave of pharmaceuticals: biologics. These drugs are based on molecules the body makes, like insulin or antibodies. Big names in the field include Genentech (1976), Amgen (1980), and Genzyme (1981). These companies were smaller and more agile than existing pharma giants, and focused on a handful of treatments instead of more blockbuster drugs. They set the groundwork for the fourth wave of pharmaceuticals—precision genetic medicine—which kicked off in the 2000s.

What genetic medicine could treat
Now those decades of work are coming to fruition, and we’re entering a “golden age” of precision genetic medicine,7 says Dowdy. At the time of writing, there are currently nine such therapies approved by the FDA for use in the US, excluding the newest Covid-19 vaccines, which are only authorized for emergency use. Now that these vaccines have made the mechanisms behind nucleic acids more mainstream, there’s even more reason to believe that they’ll skyrocket in the coming years.
There are three fields for which genetic medicine offers particularly promising innovations:
Immunizations
We know that RNA-based8 vaccines are helping vaccinate people against Covid-19. They work by instructing our cells to make the spike protein found on the SARS-CoV-2 virus which, once released into our bloodstream, spooks the immune system into action. Although the spike protein is made by a lab and not by an actual virus, the immune system makes antibodies against it—which means if it ever encounters the real thing, it’s ready.
These vaccines were authorized by the FDA so quickly for a few reasons. First, the fundamentals had been in the works for a while already. BioNTech came to the scene in 2008, and Moderna shortly after in 2010. Both of these companies were based on 40 years of work on mRNA9 as a potential vaccination pathway led by Katalin Karikó, a Hungarian-born scientist who now works for BioNTech. (CureVac, another company currently in phase 3 clinical trials for a similar mRNA-based Covid-19 vaccine, was also founded in 2010.)
Second, the sheer scale of the pandemic sped up interest in and testing for the novel vaccines. Governments were willing to pre-order vaccines, which essentially ate the costs of potential failed clinical trials—a typical barrier for speedy drug development. Also, the high number of Covid-19 cases allowed clinical trials to progress more quickly. Normally, because vaccines are administered to otherwise healthy people, they require trials including tens of thousands of people to show their safety, and it can take months or years for enough people to be exposed to prove their effectiveness. Covid-19 was spreading so quickly, however, that these trials concluded in a few short months.
In the future, though, mRNA has the potential to target so many other conditions, ranging from infectious diseases to antidotes for snake venom.
Moderna and BioNTech, the German pharmaceutical company that partnered with Pfizer to bring its Covid-19 vaccine to market, have been in the mRNA vaccine business for years. Moderna currently has 10 vaccines against infectious diseases in its pipeline. Initially, the company thought that its first vaccine to market would be one for cytomegalovirus (CMV), a herpes virus that is usually benign but, in rare cases, can cause birth defects in children. “We thought [CMV] was going to be our first vaccine with a two- to three-year clinical trial because of the prevalence of the disease,” says Robert Langer, a chemical engineer at the Massachusetts Institute of Technology who co-founded Moderna. BioNTech has four.
Each company is tackling diseases that medicine has not previously been able to protect against. Of them, the three biggest are tuberculosis, HIV, and the flu. Tuberculosis, caused by a bacterium, kills the most people of any infectious disease across the globe, taking roughly 1.4 million lives annually. HIV and the flu kill a collective 690,000 people and 646,000 people respectively each year. While there are annual flu shots for adults and tuberculosis vaccines can be given to children at a high risk of developing the illness, they’re not particularly effective and don’t reach everyone who could use a vaccine, which leaves millions of people vulnerable every year. There’s never been a vaccine for HIV.
The biggest challenge in immunizing against these conditions—and ones that these drug companies hope to solve—is that these pathogens have multiple proteins to code for, not just the one in SARS-CoV-2. But mRNA can solve this problem: It’s easy to include the code for several kinds of proteins in a single injection.
With a CMV vaccine, researchers were using four or five different kinds of mRNA to code for as many proteins. HIV, influenza, and tuberculosis presumably have similarly complex profiles. These combination vaccines will also be critical for developing a universal Covid-19 vaccine capable of protecting recipients against all the circulating variants of the virus.
Cancer therapies
Cancer is caused by genetic code that’s gone haywire. So it makes sense that using genetic material to counteract that harmful change could help resolve the cancer, too. Instead of trying to “fix” the haywire genes, though, researchers are using genetic therapies to retool our immune systems to attack cancerous cells.
One of the most sinister aspects of cancer is how it evades the immune system. Cancerous cells give off all kinds of proteins and signals—just like any other cell type—in a way that should flag them as a foreign entity your immune system would want to kill or neutralize. But cancer is a master of manipulation. “Cancer tells its microenvironments, ‘Don’t worry about it here,’” which allows sick cells to grow out of control in plain sight, says Manisit Das, a medical communications professional who has a doctorate degree in pharmacology and studied drug delivery methods at the University of North Carolina.
Personalized cancer vaccines will be a game changer, says Das. They would effectively lift cancer’s immunogenic camouflage. Like vaccines for infectious diseases, these vaccines would introduce mRNA that codes for one of the unique proteins the cancer produces, called a neo-antigen, that the body could then recognize and attack. It would also be a dramatic upgrade from current chemotherapies because, instead of flooding the body with toxic chemicals that kill cells with abandon, cancer vaccines could target only the cancer itself, leaving the rest of the body alone. Currently, several of BioNTech’s and Moderna’s other drugs in development are vaccines against cancers, although most of them are in early safety trials or animal trials.
The precursor for this technology has already been on the market for years in the form of immunotherapy treatments. The first immunotherapies reached public awareness after the FDA approved the first CAR-T (chimeric antigen receptor T-cell) therapies in 2017 for a type of leukemia. This therapy, called Kymriah and manufactured by the Massachusetts-based Novartis, takes the T-cells out of a person’s body, genetically edits them to attack cancer cells, and then re-introduces them to the person’s body. Now, there are four approved CAR-T therapies on the market, all for various blood cancers, the last of which gained approval in Feb. 2021.
Cancer vaccines, however, are just one way to fight off the disease. It’s also possible to use lipid nanoparticles to enhance the body’s own cancer-fighting abilities. “We can make lipid nanoparticles10 to encapsulate mRNA encoding tumor suppressor genes,” says Yi-zhou Dong, a pharmaceutical chemist at Ohio State University. Dong is taking a different approach to fighting cancer with nucleic medicine. These are genes that cancer cells usually turn off; by reintroducing them to cancer cells, they could get to work again. Doctors can even include more than one drug in the same lipid nanoparticle, giving the disease a one-two punch.
So far, he and his team have been able to show that this technique works well in mouse models, but they haven’t yet tested it in people. He and his colleagues are building a biotech startup that could carry out this kind of clinical work, as well as developing future therapies along these lines.
Cures or treatments for genetic diseases
When a disease starts in the genome—or in the process of turning the genome into healthy cells—it makes sense to treat it at its source. That was Timothy Yu’s line of thinking in his work as a physician-scientist at Boston Children’s.
In 2016, Yu met Mila Makovec, a sunny six-year-old with a rare condition called Batten disease. Batten disease inhibits a person from being able to get rid of cellular waste in the brain, causing toxic buildups that slowly destroy neurons. Though Mila had developed normally as a toddler, her disease caused her to slide backward; her speech, gait, and vision had become increasingly impaired. After three years of symptoms, no doctor had been able to treat her.
After analyzing Mila’s genome, Yu’s lab found the cause of her disease: a bit of extra DNA in one of the genes Mila needed to rid herself of cellular waste, which led to a mistake in the mRNA reading it, and ultimately a faulty protein. Knowing that was the culprit, Yu’s team developed a quick ASO11, which corrected the way mRNA translated her DNA, leading to the healthy proteins needed to clean out her cellular waste. It wasn’t a permanent fix—her DNA would always carry the mutation—but the ASO worked to quiet its mistakes in her brain cells’ nuclei. The process of identifying the target and creating a bespoke therapy took just over eight short months.
Through infusions in her spinal fluid every two to four months, Yu and his colleagues were able to mitigate some of Mila’s symptoms. Unfortunately, much of the disease’s damage had already been done, and Mila passed away earlier this year. “In her case, our treatment clearly bent the curve, but we were probably three years into an ongoing neurodegenerative process,” Yu says. It helped, but sadly in the end, it wasn’t quite enough.
Mila’s case, while tragic, is an important datapoint: A fatal genetic condition like hers could be successfully treated in a matter of months. In this instance, the ultimate shortcoming of the genetic therapy was connecting Mila and her family to the right doctors in time.
Mila’s particular mutation was rare, which meant Milasen—the precision genetic medicine named after her—has only worked for her so far. Yu’s research lab continues to focus on devising other kinds of ASO therapies for rare diseases. With the help of a contract research organization, he’s treating another patient named Ipek Kuzu, age three, who is living with ataxia-telangiectasia, another usually fatal genetic condition. “It takes a village to [treat her], but it doesn’t take an entire city,” he says.
For more common rare diseases—including other forms of Batten or ataxia—a single treatment may work for multiple people. Nucleic acid therapies like ASOs and RNAis12 that can correct or silence errors in genetic code are a beacon of hope for people with previously untreatable conditions. “There are lots of orphan diseases [out] there, many of which are below or off the radar screen for pharma,” says Yu. For these patients, doctors could only mitigate symptoms—there were no therapies that would allow them to intervene at the cellular level where the condition begins.
Now that’s starting to change. At the moment, there are dozens of drugs in clinical trials that can fix the way that human cells interpret broken code. That’s not just for rare orphan diseases, either; Ionis pharmaceuticals, one of the oldest genetic medicine companies founded in 1989, has drugs in the pipeline for Alzheimer’s disease, cystic fibrosis, and Hepatitis B—all of which are relatively common. One of its competitors, Arrowhead pharmaceuticals, is developing a therapy that could treat high cholesterol and cardiovascular disease, which affect tens of thousands of people worldwide.
Most of these drugs are still in phase 2 trials, which tests the drugs efficacy in patient populations; Ionis also has several ongoing phase 3 trials, including ones for ALS and Huntington’s disease. Though it’s impossible to say exactly when clinical trials would be ready for the eyes of regulatory authorities, it’s not far-fetched to assume that new treatments could be ready for the general public in the next few years.

How genetic medicine can live up to its promise
After decades of scientific progress, genetic medicine seems ready for primetime—in the world of pharmaceuticals, that means mass circulation as the state-of-the-art treatment for a variety of diseases. But to get there, the field needs better drug delivery, clear regulatory pathways, and broader accessibility for patients in need.
Getting treatment to where it’s needed in the body
For all the preliminary success of mRNAs13 and RNAis14, they really only reach three therapeutic areas in the body: muscle cells if they’re injected with a shot; liver cells if they’re delivered with an IV; and brain cells if they’re disseminated through spinal cord fluid.
But getting nanoparticles—or any vesicle containing therapeutics for that matter—to other organs is another problem to solve.
To do it, researchers will have to match the ligands15—proteins on the outside of nanoparticles—to the some 4,000 types of receptors our cells have, explains Andre Watson, a biomedical engineer.
Watson founded Ligandal, a San Francisco-based startup, nearly eight years ago to help drug companies and other research groups find the right ligands similar to those found in the body to get their particles where they need to go. There’s also some complex chemistry involved in getting ligands attached to nanoparticles, like the world’s tiniest Lego set. Though Watson and his team use a number of computational methods to try to estimate the right kinds of ligand targets for different cell-based therapeutics, the sheer number of targets the company is being asked to find is massive—and all of their work was paused to pay attention to the pandemic. At the moment, its biggest success is developing a short ligand protein that blocks the SARS-CoV-2 virus from attaching to cells. This could be a potential antiviral therapy against Covid-19, and it’s currently working on it with the US National Institutes of Health and the University of California at San Francisco.
Then, there’s the problem with lipid nanoparticles16 themselves. Sometimes, they can be too good at their job protecting their mRNA cargo. Once lipid nanoparticles get through our cell’s membranes, they get stuck in little bubbles called endosomes,17 which serve as a sorting system for incoming goods. These endosomes are also like little bubbles—and sometimes they don’t know to let the nucleic acid out to the rest of the cell. Even in circumstances where it appears lipid nanoparticles are working, “we have no idea how these things escape,” says UCSD’s Dowdy.
This is one reason why Anzalone, over at Arrowhead Pharmaceuticals, is taking an entirely different approach.“We’ve gotten really good at chemically modifying that small RNA molecule, and we do it in a way that appears to be non-toxic,” he says. Instead of trying to find ligands to attach to lipid nanoparticles, Arrowhead develops entirely new packaging for the RNA altogether by tweaking the atoms along its sugary backbone. This packaging, which is entirely proprietary, serves double duty as it both protects the actual RNA code and works as a signaling ligand.
A clear path forward with regulatory groups
In order for any drug to reach the public, it needs to get clearance from a regulatory authority like the FDA. Sometimes, individual patients with conditions that have no other treatments available can get experimental drugs either in a clinical trial or with emergency use, but it’s rare.
It’s not clear how the advent of precision genetic medicine will ultimately change the way drugs are approved. Right now, there are hundreds of lipid nanoparticles and ligands in development. Today the FDA regulates each combination of nanoparticle, ligand, and nucleic acid as its own therapy. But eventually that might not be necessary if certain lipid nanoparticles and ligands have been proven safe and effective over and over again. In that case, the only component of the therapy that would need to be evaluated would be the nucleic acid and the protein it produces or silences, which could potentially speed up the drug development time tenfold.
There’s some precedent for the FDA issuing a blanket clearance for a therapy that takes slightly different forms in different people. For example, the agency considers any product that involves directly editing someone’s genes, like in CAR-T therapy, to be a biologic—despite the fact that everyone’s T-cells will have different genetic material, and different edits may depend on their cancer. The FDA has also said that Pfizer and BioNTech won’t have to conduct separate clinical trials for future iterations of their Covid-19 vaccines built to protect against different variants; instead, the companies will have to conduct a smaller clinical trial that proves that these vaccines produce adequate antibodies.
If that’s the case, future clinical trials may be less onerous. If more precision medicine therapies are available for people with rare conditions, large clinical trials may be impractical to conduct because there are so few eligible participants. And they may not even be necessary. “Right now, it looks like monkeys are pretty good predictors of what you’ll see in the population of people,” in pre-clinical trials for genetic medicine, says Cullis. If researchers can see exactly how the nucleic acid works in primates, and the lipid nanoparticles and ligands are known to be safe, it could be even easier to bring drugs for rare diseases to market. The same may be true of therapies for more common conditions, too.
Improved access to cutting-edge treatments
The biggest barrier to the impact of any kind of new drug is the ease with which it can get into bodies in need. Drugs need to be accessible and affordable. And that’s where a lot of this work has so far fallen short.
For now, patients in need of therapies for rare diseases have to seek out research clinicians like Yu. Yu gets some of his rare-disease patient referrals from the Undiagnosed Disease Network, a research group that’s part of the US National Institutes of Health that spans the US and Canada. This network connects people with intractable health problems to clinicians who can help. But so far, even finding this network can be difficult for patients; the network has only accepted about 2,000 of the nearly 5,000 patients who’ve applied, and just three quarters of those accepted have had their genomes sequenced to be shared with their medical team. Outside of that, those with undiagnosed conditions have to search far and wide to find a doctor who can help them. Many never find one.
The treatments’ cost is also a factor. With so few gene-based therapies currently approved and on the market (aside from Covid-19 vaccines), it’s not really clear how expensive genetic medicine will be. But it’s likely they won’t be cheap. The few existing drugs on the market, such as Onpattro and Zolgensma, cost about $450,000 per year per patient or $2.1 million for life respectively (the drugs are dosed by weight, but the intent is that they would have lasting effects). Mila’s mother raised over $3 million on GoFundMe for treatments for her daughter, though the actual costs were never revealed. Insurance may cover some of these costs, but it’s not a guarantee; insurers are typically slow to cover new therapeutics.
The treatments are so expensive because they’re new, Dowdy states. Right now, there simply aren’t enough companies making nucleic acids for testing to make the process cost efficient. But, Dowdy says, it’s simply a question of scale: “Worldwide production of RNA is going up and up and up, and the cost will go down.” And even though these drugs will ultimately be more expensive per dose than traditional small molecules, RNA therapeutics only need to be administered once every three to six months. Overall, they’ll end up being cheaper in the long run for patients.
In the interim, it means that small molecule drugs still have a place in medicine. As the era of medicine they have defined comes to a close, drug companies have found ways to make them cheaply. They’ll be vitally important to treat some of these same conditions for which genetic medicine is still too expensive.
But Dowdy believes that within the next two decades, precision genetic medicine will eclipse other types of treatments once and for all. Within a routine doctor’s visit, patients of the future could receive the shots or injections they’d need to manage their health for the next four to six months—maybe even a year. The fourth wave of pharmaceuticals is imminently upon us.
Correction: A previous version of this article incorrectly stated which drug costs $2.1 million. That drug is Zolgensma, not Spinraza.
