Two industrial titans are duking it out over a technology that could make electric cars mainstream
The high-tech patent wars have spread to a new front, engaging two of the world’s largest industrial companies in a multibillion-dollar court battle over lithium-ion batteries. At issue is a battery chemistry that, while little known to the public, many experts believe currently holds the best chance of electric cars penetrating the mass market.


The high-tech patent wars have spread to a new front, engaging two of the world’s largest industrial companies in a multibillion-dollar court battle over lithium-ion batteries. At issue is a battery chemistry that, while little known to the public, many experts believe currently holds the best chance of electric cars penetrating the mass market.
The case pits Germany’s BASF, the world’s largest chemical company, against Belgium’s Umicore, one of the biggest makers of battery materials. Filed by BASF on Feb. 20 in US federal court in Delaware, the lawsuit—which has been reported only by patent blogs—accuses Umicore of selling a key battery component to which BASF holds an exclusive license. It also alleges that Umicore threatened to sue firms if they gave their business to the German company. The suit seeks billions of dollars in alleged damages.

In addition to the pair of European giants, the case tees up a potential collision between two lithium-ion titans with rival patents for the technology at the heart of the dispute—3M Corp., an industrial giant itself, and Argonne National Laboratory, a US government lab that is a co-complainant with BASF.
The case, if it is not settled informally before trial, will require jurors to make sense of nuanced interpretations of the arcane atomic structure of battery electrodes. Specifically, they will hear dueling versions of what goes on at nano-scale when you charge and discharge a battery containing “NMC,” a positive electrode—or cathode, which is the nervous system of a battery—containing nickel, manganese, and cobalt.
To make their case, lawyers for BASF and Argonne are likely to describe a battle under way among major carmakers to launch next-generation electric cars in the 2017-2020 time frame—Tesla, GM, BMW, and possibly Apple and Virgin among them. They will allege that Umicore has effectively locked BASF out of the contest.
“BASF has lost out on billions of dollars of potential revenue from selling [NMC] materials because of Umicore’s misrepresentations to major purchasers in the [NMC] materials market,” BASF and Argonne claim in the lawsuit. “In addition, BASF has lost the ability to compete as a supplier for electric vehicle platforms expected to launch in 2016 and 2017.”
In recent years, gigantic patent battles have roiled the market for tablets and smartphones, pitting Apple against Samsung, Nokia, Microsoft, and almost anyone else of consequence. Combat also has been rife in the market for gaming devices. The total number of such suits plunged last year, possibly as a result of a US Supreme Court ruling that made it harder to win. But large cases continue to be filed.
With their lawsuit, BASF and Argonne underline the rising economic stakes in batteries. While technological advances in batteries have been slow to come—thus stunting the expected appeal of electric cars—lithium-ion has nonetheless grown into a respectable $18-billion-a-year industry, according to Navigant, a clean energy research firm. This is because of the stunning popularity of smart phones and tablets, plus sales of the electric Tesla S, the GM Volt, and the Nissan Leaf, all of them requiring lithium-ion batteries. Continued double-digit annual growth should push the lithium-ion battery market to about $38 billion by 2020, Navigant says.
The lawsuit names Umicore and one of its customers—Japan’s Makita Corp., a toolmaker. It cites laboratory tests allegedly showing that Makita tools contain NMC invented by Argonne, and says that the Japanese company obtained the cathode material from Umicore. It says that neither Umicore nor Makita have licenses for Argonne’s NMC.
In a statement emailed to Quartz, Umicore disputed BASF’s assertions and said it intends to fight the lawsuit. Makita did not respond to a phone message left at its La Mirada, California, office. 3M declined to comment. BASF lawyer Brian Farnan did not respond to emails. Argonne said it could not comment beyond what it said in the lawsuit.
Argonne was first, but 3M cornered the non US-market
The competing NMC patents go back a decade and a half. The first was filed in June 2000 by Michael Thackeray, a South African-born researcher working at Argonne. Ten months later, 3M filed a competing patent application on behalf of Jeff Dahn, a researcher at Dalhousie University in Halifax, Nova Scotia.
Thackeray’s patent is good only for the US—at the time it was filed, neither he nor Argonne’s lawyers expected the NMC to take on the industry importance that it has, and so they sought to economize by filing for no international patents. But 3M, calculating differently, obtained patents in the US, plus the world’s most important manufacturing markets—China, Japan, and South Korea.
Over the years, 3M has aggressively defended the overseas patents, threatening and filing lawsuits for alleged infringement against Sony, Matsushita, and Sanyo, and winning settlements in each case. The details of the settlements are sealed, but the outcomes appeared to validate 3M’s claims.
3M has apparently not filed similar lawsuits in the US. Now, Argonne, teamed up with BASF, has taken the offensive.
A ruling against BASF and Argonne could have important implications outside the Umicore-Makita issue. For instance, one of the largest customers for NMC is General Motors, which uses it in the Chevy Volt. Argonne has stated publicly that the Volt’s battery contains its version of NMC. But Dahn has said that the Volt battery contains his work, and not Argonne’s. If BASF and Argonne lose their case against Umicore, the legal question could then arise of whose cathode, chemically speaking, is actually in the Volt.
General Motors referred questions to LG Chem, which makes the Volt cathode. LG’s NMC is produced under a license with Argonne.
We will hear rare electrochemical nitty-gritty
The dual challenges of lithium-ion batteries are to obtain more energy at a lower cost. The current standard—a 35-year-old battery formulation in which the cathode is made of lithium cobalt oxide—packs a lot of dense energy, but is expensive and also more volatile than almost any commercial rival.
Though it has its own challenges, the NMC may have the best chance to beat lithium cobalt oxide, while performing well on the other metrics of energy and cost, many experts say. Among the reasons are that the cathode contains less cobalt, which is expensive, and also is less volatile, and thus safer for use in electric cars.
Over the years, Thackeray and Dahn have bickered over the precise atomic structure of the NMC—is it a saucy amalgam of metals, or a “solid solution”(Dahn’s position), or is it a more structured composite with a discernible chemical architecture (Thackeray’s)?
Until now, their dispute had seemed largely academic, obscure, and possibly immaterial. But now, who a jury determines is right may end up as the pivotal factor in the BASF/Umicore case. (Quartz reached out to Dahn and Thackeray; both declined to comment on the suit.)
In his ground-breaking patent, Thackeray had the central thesis that if you built up the amount of lithium in the NMC—if you added about 10% more to the cathode—you achieved a big increase in capacity. To understand what he meant, and what’s at the heart of the legal dispute, it’s useful to look both at how lithium-ion batteries work, and specifically at how Thackeray’s cathode manages this jump.
All lithium-ion batteries have three basic parts—two electrodes and, between them, a facilitator called electrolyte. When you plug in your smartphone, lithium stored in the cathode begins to shuttle to the other electrode, called the anode. Then, when you unplug your fully charged phone and start to use it, the lithium begins to shuttle back from the anode to the cathode.
One of battery science’s objectives is to get as much of the lithium as possible to shuttle, because that creates more energy. But no matter what, you cannot put all of it in motion, because, emptied out of so much material, the cathode will collapse in on itself. A rule of thumb with the standard lithium cobalt oxide cathode is that you can put about half of the lithium into the shuttling motion.
Thackeray’s formulation claimed to beat that rule of thumb. It starts with two compounds—the NMC itself, plus a chemistry called Li2MnO3. Visually, both the NMC and Li2MnO3 resemble stripped-down houses. The floors and ceilings are made of oxygen atoms, and the walls are nickel, cobalt, and manganese. Scientists call this framework a lattice. Because the lattices of the NMC and the Li2MnO3 are similar, you can easily integrate the two at the nanoscale.
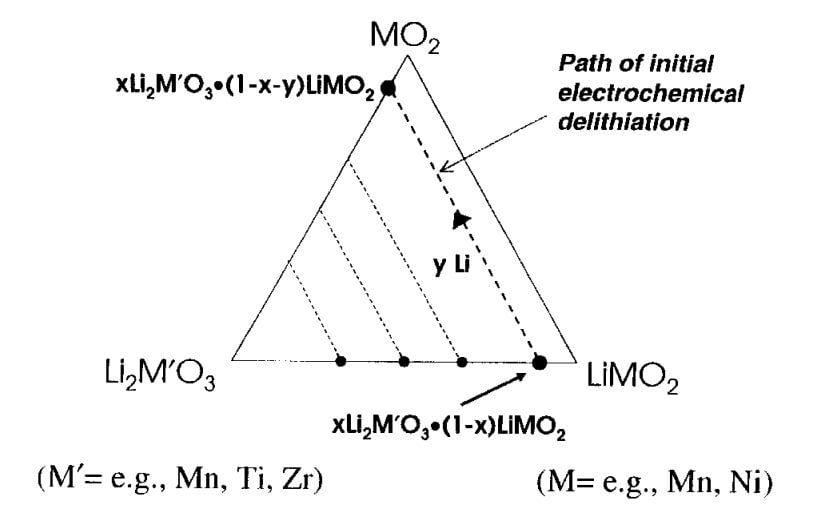
And that’s what Thackeray did–he shifted the Li2MnO3 into the NMC—at which point two things happened: The manganese and lithium propped up the NMC like pillars, adding to its sturdiness, and the extra lithium increased its capacity.
The former phenomenon (the sturdiness) was responsible for the latter—the capacity. This was because, with the double lattice at work, you could now shuttle 60% or 70% of the lithium without the cathode collapsing.
And here also is where the distinction comes between the rival cathodes. Thackeray called his invention a “layered-layered” or “composite” cathode. The atoms of the various metals were clearly concentrated in clumps throughout the electrode, he said.
But Dahn called his version a “solid solution.” The same metals were present of course, but they were more or less evenly arrayed, he said.
What’s the real difference? We’ll hear in court.